
When these two conditions are met, the equation is said to be balanced. How to balance chemical equation by ion-electron method. 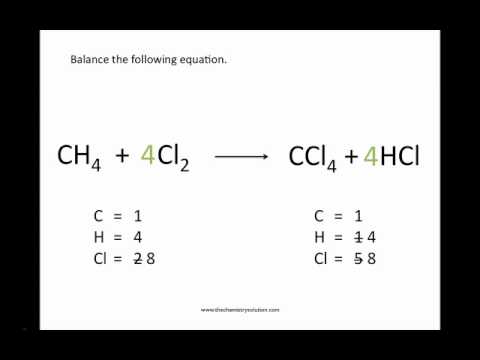
For specifically calculating the oxidation number using online tools, we provide you oxidation number calculator to get the job done easily with few clicks. Also the sum of the charges on one side of the equation must be equal to the sum of the charges on the other side. The balancing chemical equations calculator becomes the best option which ensures the correctness of the final equation. 5.3: Precipitation Reactions and Solubility Guidelines is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. Solubility rules are used to predict whether some double-replacement reactions will occur. This means that a chemical equation must have the same number of atoms of each element on both side of the equation. A precipitation reaction is a double-replacement reaction in which one product is a solid precipitate. The Law of Conservation of Mass states that mass is neither created nor destroyed in an ordinary chemical reaction. 
If the elements in a chemical formula are properly capitalized, the smart case converter leaves them as you have typed.Ī balanced chemical equation accurately describes the quantities of reactants and products in chemical reactions. The general forms of these five kinds of reactions are summarized in Table 7.10.1, along with examples of each. You may notice that in a complete ionic equation, some ions do not change their chemical form they stay exactly the same on the reactant and product sides of the equation. Most chemical reactions can be classified into one or more of five basic types: acidbase reactions, exchange reactions, condensation reactions (and the reverse, cleavage reactions), and oxidationreduction reactions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
